Introduction
|
Vascular EDS is a rare yet fatal disease with sudden death often resulting from organ, uterine, or arterial rupture (1). Individuals affected with Vascular EDS produce less functional collagen as a result of the mutation in COL3A1. Often amino acid substitution mutations are responsible for Vascular EDS. This substitution results in faulty protein folding resulting in the collagen deficit (2). There is no known cure for Vascular EDS nor treatments highlighting the importance of further research on this disease. Interestingly, for individuals affected with Vascular EDS, 18% of males die from vascular rupture before the age of 20, whereas only 7% of females die of the same cause by the same age leading to the male-biased lethality of this disease (1) . |
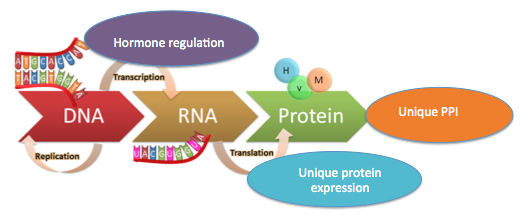
Figure 1: There are different steps throughout the central dogma where differences between males and females may be contributing to the overall male-biased lethality of Vascular EDS.
|
My goal is to identify factors that influence the male-biased lethality of those affected with Vascular EDS during adolescence. Gender differences in type III collagen production have been shown in mouse models: collagen levels are similar between males and females until puberty, when males exhibit a drastic increase in collagen production (3). I hypothesize that hormone receptor activity during adolescence leads to differential protein expression and altered protein interactions in male and female hearts, contributing to the male-biased lethality of Vascular EDS.
Aim 1: Hormone regulation
|
In order to investigate the hormonal differences between the sexes I will look at the activity of testosterone and estrogen receptors when bound to their respective ligands. In order to look at this interaction I will utilize Chromatin Immunoprecipitation assay followed by next generation sequencing (ChIP-Seq), a good method for investigating the interactions between proteins and DNA in the cell, on heart tissue from male and female mice. Antibodies against testosterone and estrogen will be added and desired complexes will be precipitated out. The resulting DNA bound to estrogen and testosterone will be sequenced with next generation sequencing to determine what proteins are under the hormonal regulation of estrogen and testosterone receptors. |
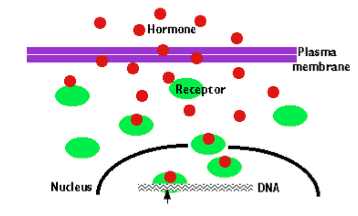
Figure 2: Hormones and their receptors acting as transcription factors. Hormone receptors can act as transcription factors when they are bound to their respective ligands.
|
|
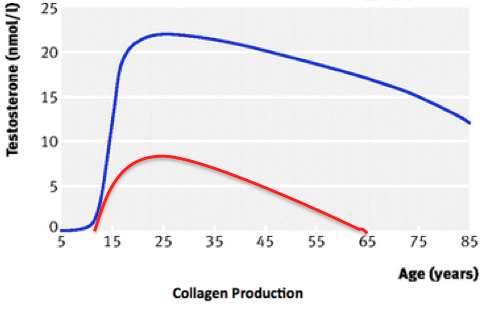
Figure 3: Testosterone levels throughout development with males in blue and females in red shows a peak in testosterone during male adolescence
|
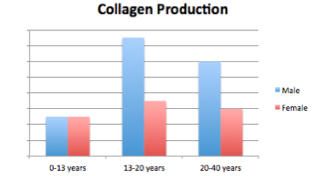
Figure 4: Collagen production is relatively similar until adolescence when males exhibit a drastic peak in collagen. This difference is maintained through adulthood.
|
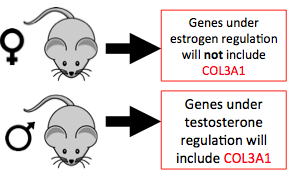
Figure 5: Due to the peak in testosterone and collagen during male adolescence I predict that COL3A1 is under hormonal regulation.
|
Due to the peak of collagen during male but not female adolescence (Figure 4), I expect COL3A1 to be under the transcriptional regulation of testosterone receptors but not estrogen receptors. In addition to COL3A1, I expect other genes that share GO terms with COL3A1, including extracellular matrix components, to also be under hormonal regulation. If COL3A1 and other proteins with similar gene ontology are shown to be under hormonal regulation there is the potential to use endocrine therapy to modulate hormone levels to change gene expression and potentially disease outcome to eliminate the male biased lethality of Vascular EDS.
Aim 2: Unique Protein expression
|
After identifying which proteins are under hormonal control it will be important to determine what proteins are expressed in male and female hearts at different points during development. Healthy mice will be compared to diseased mice in this experiment, Mice that are homozygous knockouts for COL3A1 are not compatible with life and die within 48 hours of birth (4). Therefore,we will be using COL3A1 haploinsufficient mice which mimic the phenotypes of humans with Vascular EDS. Quantitative Mass Spectrometry will be performed at 2 months, 14 months, and 18 months of mouse development, corresponding to childhood, adolescence, and adulthood in male and female heathy and diseased mice in order to determine what proteins are expressed and their expression level. |
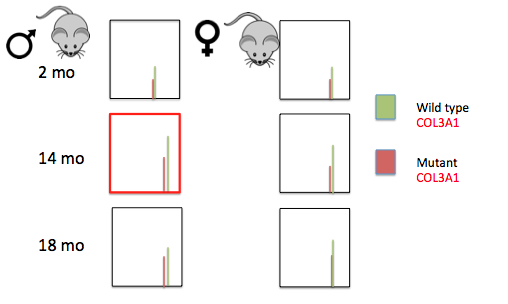
Figure 4: Expression of COL3A1 in male and female mice at time points corresponding to childhood, adolescence, and adulthood. Healthy (green) and COL3A1 haploinsufficient mice (red) were used in this experiment.
|
We expect Mass Spectrometry for COL3A1 to show peaks during adolescence when testosterone peaks, with higher levels seen in males overall. In addition, mutants will have lower levels of COL3A1 overall, consistent with the Vascular EDS phenotype. I predict that the proteins that are expressed in male adolescent mice, indicated by the red box in figure 4, will be important in the male-biased lethality of Vascular EDS. Furthermore, I believe that these proteins will exhibit similar GO to COL3A1, including extracellular matrix components, and importance in heart and organ formation. Lastly, I believe that the genes shown to be under hormonal regulation in aim 1, will correspond to the proteins that are up-regulated during adolescence in this aim.
Aim 3: Unique Protein interactions
|
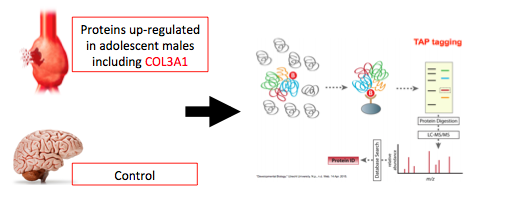
In addition to unique proteins being expressed in the hearts of adolescent male mice, we expect unique protein interactions to occur in the hearts of male adolescent mice which may contribute to male-biased lethality. In order to identify unique protein interactions during adolescence a Tap-Tag assay will be performed. Proteins identified in 14 week old mice from aim 2 will be used as baits in the Tap-Tag assay. Brain tissue sample will be used as a control. Tap-Tag assays are useful for identifying a wide range of protein-protein interactions beyond the bilateral interactions that occur in the nucleus which Yeast-2-Hybrid assays are able to identify (5). Because COL3A1 is found in the extracellular matrix and we expect many different interaction partners a Tap-Tag will provide the most relevant information.
|
Figure 5: Proteins up-regulated during male adolescence in diseased and healthy male mice will be used in a Tap-Tag assay to identify their interaction partmers
|
|
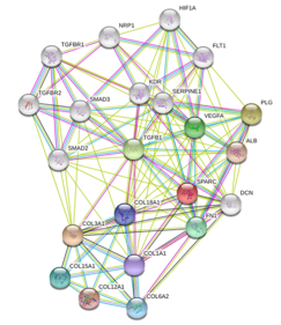
As shown in the proteomics analysis, SPARC, an important interaction partner of COL3A1, is responsible for cell growth. In order to gather more information on the relationship between these two proteins, I ran them in parallel on STRING to determine overlap in interaction partners. SPARC, shown in red, and COL3A1, shown in purple, turn out to have an extensive network of interaction partners as seen in figure 6. This is an example of what results I hope to obtain from the Tap-Tag assay when I look beyond bilateral protein-protein interactions. Once protein interactions that occur during male adolescence have been identified we can compare the interactions that occur in healthy adolescent males to those that occur in the COL3A1 haploinsufficient mice. I predict that the protein interactions that occur in diseased mice during adolescence may play a role in the male-biased lethality of Vascular EDS and can serve as future targets of therapies that attempt to modulate these interactions |
Figure 6: Overlap of Protein-Protein interactions between COL3A1 and SPARC produced by STRING database.
Future Directions
|
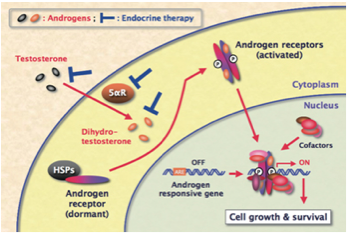
Endocrine therapy can be used to add, block or remove hormones (6). Figure 7 demonstrates how endocrine therapy can be used to block hormones and the resulting transcription factor activation. If COL3A1 is shown to be under hormonal regulation I hypothesize that using endocrine therapy to up-regulate the transcriptional activity of testosterone receptors may improve the outcome for male patients with Vascular EDS. Males produce more collagen during adolescence because it is needed for their development. Thus, the Vascular EDS phenotype which reduces the amount of functional collagen produced due to improper folding may be more detrimental in males because they need higher levels overall. I predict that if COL3A1 is under hormonal regulation and endocrine therapy is used to up-regulate testosterone receptors more collagen will be produced leading to an overall increase in functional collagen for individuals with Vascular EDS. As a result, if lack of function collagen is what is leading to the arterial and organ ruptures in Vascular EDS, I hope that this increase in collagen production will improve the outcome for males with this disease.
|
Figure 7: Endocrine therapy is being used to block the activity of androgen receptors as transcription factors by preventing testosterone from activating the androgen receptors. As a result androgen responsive genes are not transcribed.
|
It is important to determine how gender hormone differences regulate protein expression in hollow organ walls, what proteins are expressed in hollow organ walls during different time points in development as a result of gender hormone differences, and how these proteins interact with each other differentially in each gender. This information will provide more details on the mechanism of male-biased lethality of Vascular EDS and ultimately a starting point for drug targets and potential treatments. Beyond modulating COL3A1 levels with endocrine therapy, specific proteins and protein interactions that are shown to be up-regulated in male adolescent diseased mice may serve as potential drug targets in an attempt to reduce the male-biased lethality of Vascular EDS.
References
1) Ong, KT, J. Perdu, J. Backer, E. Bozec, P. Collignon, J. Emmerich, AL Fauret, JN Feissinger, DP Germain, G. Georgesco, JS Hulot, A. De Paepe, H. Plauchu, X. Jeunmairte, S. Laurent, and P. Boutouyrie. "Result FEffect of Celiprolol on Prevention of Cardiovascular Events in Vascular Ehlers-Danlos Syndrome: A Prospective Randomized, Open, Blinded-endpoints Trial." Lancet (2010): 1476-484. PubMed. U.S. National Library of Medicine, 30 Oct. 2010. Web. 20 Feb. 2015. <http://www.ncbi.nlm.nih.gov/pubmed/20825986>.
2) Pepin, Melanie G. "Ehlers-Danlos Syndrome Type IV." GeneReviews. U.S. National Library of Medicine. Web. 12 May 2015. <http://www.ncbi.nlm.nih.gov/books/NBK1494/>.
3) Markova, Marina, Joeseph Zeskand, Benjamin McEntee, Jay Rothstein, Linda Siracusa, and Serigo Jimenez. "A Role for the Androgen Receptor in Collagen Content of the Skin." Journal of Investigative Dermatology (n.d.): 1052-056. Nature.com. Nature Publishing Group. Web. 25 Feb. 2015. <http://www.nature.com/jid/journal/v123/n6/full/5602593a.html>.
3)http://www.ncbi.nlm.nih.gov/pubmed/20587693
5) Grimm, Bernhard. "High-throughput Analysis of Bilateral Protein-protein Interactions Using the Yeast Two Hybrid System." Endocytobiosis and Cell Biology. Web. 13 May 2015. <http://zs.thulb.uni-jena.de/receive/jportal_jparticle_00252058>.
6) "Hormone Therapy and Breast Cancer." WebMD. Web. 13 May 2015. <http://www.webmd.com/breast-cancer/hormone-therapy-overview>.
1) Ong, KT, J. Perdu, J. Backer, E. Bozec, P. Collignon, J. Emmerich, AL Fauret, JN Feissinger, DP Germain, G. Georgesco, JS Hulot, A. De Paepe, H. Plauchu, X. Jeunmairte, S. Laurent, and P. Boutouyrie. "Result FEffect of Celiprolol on Prevention of Cardiovascular Events in Vascular Ehlers-Danlos Syndrome: A Prospective Randomized, Open, Blinded-endpoints Trial." Lancet (2010): 1476-484. PubMed. U.S. National Library of Medicine, 30 Oct. 2010. Web. 20 Feb. 2015. <http://www.ncbi.nlm.nih.gov/pubmed/20825986>.
2) Pepin, Melanie G. "Ehlers-Danlos Syndrome Type IV." GeneReviews. U.S. National Library of Medicine. Web. 12 May 2015. <http://www.ncbi.nlm.nih.gov/books/NBK1494/>.
3) Markova, Marina, Joeseph Zeskand, Benjamin McEntee, Jay Rothstein, Linda Siracusa, and Serigo Jimenez. "A Role for the Androgen Receptor in Collagen Content of the Skin." Journal of Investigative Dermatology (n.d.): 1052-056. Nature.com. Nature Publishing Group. Web. 25 Feb. 2015. <http://www.nature.com/jid/journal/v123/n6/full/5602593a.html>.
3)http://www.ncbi.nlm.nih.gov/pubmed/20587693
5) Grimm, Bernhard. "High-throughput Analysis of Bilateral Protein-protein Interactions Using the Yeast Two Hybrid System." Endocytobiosis and Cell Biology. Web. 13 May 2015. <http://zs.thulb.uni-jena.de/receive/jportal_jparticle_00252058>.
6) "Hormone Therapy and Breast Cancer." WebMD. Web. 13 May 2015. <http://www.webmd.com/breast-cancer/hormone-therapy-overview>.